What battery systems are we expecting in the future?
As the demand for residential and commercial storage systems continues to grow, batteries are becoming increasingly important in our everyday work. But how sustainable are the batteries? And what cell types are there? How will the market for batteries develop in the future?
Recyclability of batteries
If you look at the raw materials used and the production processes of a battery, it does not seem to be sustainable or environmentally friendly. This is because a battery contains critical and scarce raw materials, such as cobalt, lithium or manganese. The mining and production of these materials can have significant social and environmental impacts in the countries where they are produced.
In my opinion, batteries are environment friendly as they reduce our dependence on fossil fuels. Moreover, they are becoming increasingly sustainable. Readily available and non-critical resources such as sodium are used in their manufacturing. The extraction of critical materials is also becoming environment friendly as we learn from our mistakes.
For example, did you know that lithium mining in Chile has become more environmentally friendly? In the past, the brine (which contained the lithium) was pumped to the surface and stored in huge basins until the water evaporated. This caused the groundwater level to drop. Now the lithium is extracted directly from the brine and the brine is pumped back into the ground.
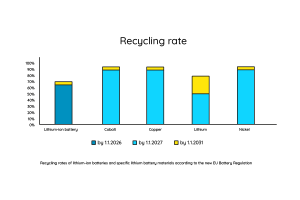
What happens when the battery runs out? Can battery modules be recycled? Over 96% of the materials used in a battery module can be recycled. In the past, lithium batteries were mostly incinerated and then crushed to recover some of the raw materials such as cobalt, nickel and copper. Now a new process, known as mechanical hydrometallurgy, is being widely used. It looks like this:
- The battery module is ground
- Using several complex sorting processes, the raw materials are separated
The process is market-ready and suitable for industrial scale. Several companies are already using it successfully. A German car manufacturer wants to build a plant that can recycle 2500 tonnes of battery modules using this process. And this is just the beginning!
According to the EU Battery Regulation, at least 70% of all battery modules should be recycled by 2030.
Let’s take a closer look at the battery cell. It plays a central role in both storing and releasing the energy. Basically, battery cells are built in one of these designs:
- Cylindrical (classic round cells)
- Prismatic
- Pouch (also known as coffee bag cell)
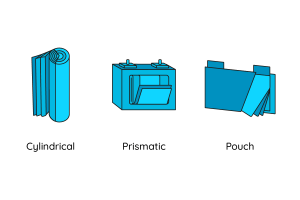
Battery structures
Each design has its advantages and disadvantages. For example, the cylindrical shape enables a very high volumetric energy density at cell and module level. However, the round shape cells fill the space very inefficiently. In terms of energy density, the battery cell in a pouch structure can keep up with the cylindrical shape. The space utilisation is efficient, but the pouch structure is very flexible and therefore inflates under pressure. Prismatic battery cells have a low energy density, but at the same time are very robust and make good use of space.
Current battery systems and cell technologies
Lithium-ion (Li-ion) batteries are the most common type of batteries used in stationary systems today. Li-ion battery systems are well scalable, highly durable, require low maintenance, have high energy density, can withstand high/low temperatures and are less susceptible to humidity than other battery systems. In order to increase the safety of Li-ion battery cells and to avoid major damage/risks, the following elements are installed:
- PTC (Positive Temperature Coefficient) element: It breaks electrical contact with the cell when it heats up and can prevent the cell from overheating further.
- CID element (Current Interrupt Device): It breaks electrical contact with the cell in the event of overpressure.
The lithium-nickel-cobalt-aluminium-oxide (NCA) battery has been around since the late 1990s. It offers more specific energy and high performance. NCA battery cells require special monitoring mechanisms in electric cars to ensure greater safety. NCA battery cells are expensive to produce, despite the low use of cobalt in it. They are not widely used in the automotive industry.
Lithium-nickel-manganese-cobalt-oxide (NMC) battery cells can be produced with different cell properties by varying the proportions of nickel, manganese and cobalt. Less cobalt means lower cost. More nickel increases the energy density. A larger proportion of manganese increases the specific power. This flexibility is a great advantage of these batteries.
Lithium iron phosphate (LFP) battery cells offer higher intrinsic safety and power density. The element groups used for cell production have better availability, which makes the production process much easier. Disadvantages are faster self-discharge and lower energy density compared to NCA and NMC cells. Further comparisons:
| LFP | NCA | NMC | |
| Energy density | Low | High | Middle |
| Fast charging | High | Low | Middle |
| Power output | Low | High | Middle |
| Life span | High | Middle | Middle |
| Cost | High | Middle | Middle |
| Safety | High | Low | Middle |
| Availability of materials | High | Low | Low |
This is what the future of the battery market is going to look like:
The battery market is poised for growth – 214,000 residential storage systems and around 3900 commercial storage systems will be added in 2022 alone. Estimates suggest a demand of 2035 GW in 2030. Much of this will be used for electromobility.
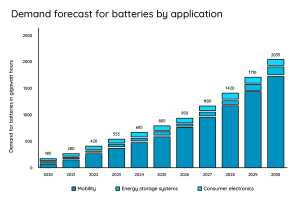
In order to meet this demand, research is being carried out to find cost-effective and sustainable alternatives to the lithium-ion battery. A small overview of the projects:
Cerenergy – a low-cost alternative to Li-ion batteries?
The Fraunhofer Institute for Ceramic Technologies and Systems IKTS has set up a joint venture. The cell technology will be marketed under the brand name “Cerenergy “. This new technology is free from lithium, cobalt, graphite and copper. It can therefore reduce raw material shortages and supply chain problems.
According to the researchers, the production costs are about 40 % lower than the costs for lithium-ion batteries. The main components of the Cerenergy battery are sodium chloride or Na/NiCl2 and Na/S batteries. The newly developed Cerenergy ACB60 battery packs are said to have a longer life span and to be able to deliver the stored energy for a longer period.
CATL – Chinese manufacturer of sodium-ion (Na-ion) batteries (production from 2023)
In 2023, the Chinese manufacturer Contemporary Amperex Technology (CATL) will introduce a new Na-ion battery. Initially, Na batteries will replace lead-acid batteries in fuel-based cars, but in the medium or long term they will also be used in electric cars. This battery technology impresses with its super-fast charging and compatibility, which is important for e-cars. CATL has also developed a combination of lithium-ion and Na-ion cells. It is suitable for the manufacturer’s cell-to-pack technology and can be used without battery modules in electric cars.
Aluminium-sulphur battery (Massachusetts Institute of Technology (MIT), USA)
The aluminium-sulphur battery technology was developed by an international team of researchers at the Massachusetts Institute of Technology (MIT) in the USA. In addition to aluminium and sulphur, chloraluminate salt, which acts as an insulator, is used here to store energy. Numerous tests have shown that this battery technology has a high charging speed. In addition, the cost is 1/6 that of lithium-ion batteries. But the biggest advantage is that there is no fire hazard.
Conclusion
I think we have come a long way in terms of the sustainability of battery systems. If I compare resource extraction and recycling technologies now with those of a few years or decades ago, the industry has made great progress. In terms of cell construction and cell chemistry, we have reached a very high standard of safety and performance. In the future, we will need a mix of different battery technologies to provide the right solution for every application.
In summary:
- Battery systems are becoming increasingly sustainable.
- There are 3 different cell types of batteries: cylindrical, prismatic and pouch.
- At present, Li-Ion batteries dominate the market and among them especially: NMC, NMA and LFP cell chemistries.
- Li-Ion alternatives are becoming increasingly important.


